Diagnostics for medication intolerance
Medication allergies are common!
8 % of patients treated as outpatients and 20 % of hospitalised patients show adverse drug reactions. In 80 % of cases these are not allergies but rather adverse ‘side effects’ of the active substance. Nevertheless, the fraction of true medication allergies is still considerable at 20 %. Unlike other side effects, an allergy involves inflammatory reactions that are induced by interactions between medication components or metabolites and allergen-specific antibodies or immune cells. In principle, every preparation (even those that do not require a prescription!) can cause an allergy.
An allergic reaction to medications cannot be predicted. Patients with known atopic disposition do not suffer more often from medication allergies. On the other hand, medication allergies can occur in patients with a completely normal allergy history. It is only known that patients with systemic viral infections react more frequently to antibiotics (e.g., ampicillin with mononucleosis).
Manifestations of medication allergy
The symptoms of a medication allergy are determined by the particular pathomechanisms which are divided into 4 types according to Coombs and Gell. Allergy types I and IV dominate (see table). A special form is the pseudoallergy which has symptoms that resemble those of type I allergies but which is not triggered by IgE antibodies and is therefore not detected by IgE or epicutaneous testing.
The symptoms of a medication allergy depend on the allergy type present.
There are allergies of type I to type IV.
Type I (IgE mediated): Urticaria, rash, weals, erythroderma, rhinitis, asthma and anaphylaxis
Type II (IgG/complement mediated): Granulocyto-paenia, erythrocytopaenia, lymphocytopaenia
Type III (IgM/complement mediated): Vasculitis, arthritis, glomerulonephritis
Type IV (T lymphocyte mediated): Skin eczema, hepatitis (liver values ↑), nephritis, flu-like symptoms
Diagnosis of medication allergy of type I and type IV
The diagnostics include a medical history, clinical results, skin tests and the laboratory diagnostics. Provocation tests are, with the exception of ASA intolerance, are usually omitted with medication allergies.
Depending on the symptoms (see above) and primarily on their development over time (< 24 hours usually type I; > 24 hours more likely to be type IV), the basophil degranulation test (BDT) and/or the lymphocyte transformation test (LTT) will be done in the laboratory.
How do these test work?
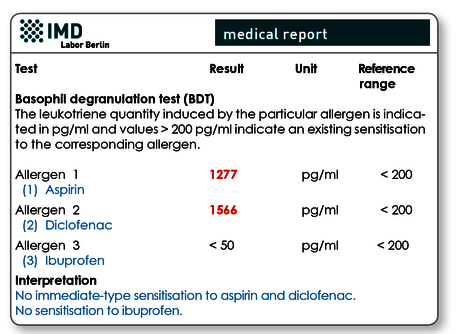
These test procedures are in vitro provocation tests, that is, the blood cells of the patient (lymphocytes for the LTT or basophils for the BDT) are ‘provoked’ with the suspected allergen in the laboratory. The BDT (synonyms: CAST test, basophil activation test) measures the release of leukotrienes and the LTT measures lymphocyte activation as ‘evidence’ that there is an individual sensitisation.
In both laboratory tests there is the option of testing active substances that are available in the laboratory but native substances (e.g., tablets, capsules, injection solutions) can also be sent with the blood sample to the laboratory.

IgE determination is less helpful
Determining the specific IgE in the serum is common for inhalation allergies, but is not really helpful with suspected medication allergies, because it can only be used with a few active substances (testing of native preparations is not possible) and type IV allergies are not detected. The exceptions are, if required, IgE antibodies to penicillin and penicillin derivatives (rarely false positive) which are only moderately sensitive, however (often false negative). The following applies therefore: a positive IgE test is an indication of sensitisation but a negative test does not rule one out.
Type II and type III allergies are difficult to verify
IgG- and/or IgM-mediated type II and type III allergies cannot be proven using laboratory diagnostics. Allergen-specific IgG and IgM antibodies cannot be detected, because they are not formed against the medication itself but rather against cellular structures modified by the medication. These are not accessible to allergy testing. With sudden cytopaenia in the blood count, a type II allergy should be considered and with vasculitis, sudden onset arthritis or glomerulonephritis a possible type III allergy should be considered.
What are pseudoallergies?
Pseudoallergies (idiosyncratic reactions), which are clinically very similar to type I allergies, are also possible. In the majority of cases these are caused by the active substance or an additive or preservative ‘switching on’ the activation cascade of mast cells without a prior IgE-receptor-mediated activation (therefore ‘pseudo…’). Pseudoallergies can be verified with laboratory diagnostics using the basophil degranulation test (BDT).
Material
BDT (per medication): 2 ml heparin blood or EDTA
LTT (per medication): 5 ml heparin blood plus (one-off)
5 ml serum
Sample receipt within 24 hrs has to be ensured. The sample should be stored and transported at room temperature. Within the Berlin city area, we offer a courier service (+49 (0)30 7701- 250). For collections beyond Berlin, please contact our complimentary courier service (+49 (0)30 77001-450)
The following active substances are available in the laboratory for testing:
Antibiotics
Amoxicillin, ampicillin, cefazolin, ceftriaxon, cefuroxime, cephalosporin, ciprofloxacin, clarithromycin, clavulanic acid, clindamycin, doxycycline, erythromycin, levofloxacin, moxifloxacin, penicillin g, penicillin v, rifampicin, sulfamethoxazole, trimethoprim, tetracycline
Pain relief
Aspirin/acetylsalicylic acid, diclofenac, ibuprofen, indometacin, mefenamic acid, metamizole, paracetamol, phenylbutazone, propyphenazone, tramadol
Anaesthetics
Lidocaine, mepivacaine, articaine, prilocaine, ubistesin
However in the case of local anaesthetics we recommend examining the native preparation rather than the active substance due to possible additives. An ampoule together with the blood would have to be sent in.
Muscle relaxants
Atracurium, mivacurium, pancuronium, propofol, rocuronium, suxamethonium, vecuronium,
Others
Bisoprolol, Ramipril, Chlorhexidine
Preparations not listed here should be sent with the blood samples (a blistered tablet or capsule, an ampoule; please do not send filled containers due to the risk of contamination).
Invoicing
Pleas obtain the costs for the BDT and LTT test from the pdf document.
Would you like to see a presentation on the matter?
In our video archive, you can find a free presentation on this topic. Access is free und possible without prior registration.
Literature
- Jurado-Palomo J. Et al. (2010) Use of the lymphocyte transformation test in the diagnosis of DRESS syndrome induced by ceftriaxone and piperacillin-tazobactam: two case reports. J Investig Allergol Clin Immunol.;20:433–6.
- Kano Y. (2007) Utility of the lymphocyte transformation test in the diagnosis of drug sensitivity: dependence on its timing and the type of drug eruption. Allergy; 62: 1439–44.
- Merk HF (2005) . Lymphocyte transformation test as a diagnostic test in allergic contact dermatitis. Contact Dermatitis; 53:246–51.
- Merk HF. (2005) Diagnosis of drug hypersensitivity: lymphocyte transformation test and cytokines. Toxicology, 209:217–20. Review.
- Nyfeler B. et al. (1997) The lymphocyte transformation test for the diagnosis of drug allergy: sensitivity and specificity. Clin Exp Allergy, 27:175–81.
- Pichler WJ. Et al. (2004) The lymphocyte transformation test in the diagnosis of drug hypersensitivity. Allergy; 59:809–20. Review.
- Piñero Saavedra MC et al. (2011) Usefulness of lymphocyte activation test in atorvastatin hypersensitivity. J Investig Allergol Clin Immunol., 21:574–5.
- Romano A. et al. (2011) Diagnosis and management of drug hypersensitivity reactions. J Allergy Clin Immunol., 127:67–73.
