Laboratory Diagnostics of chronic inflammatory multisystemic diseases
Chronic inflammatory diseases are on a rise
Chronic inflammatory diseases are multisystemic diseases that primarily or secondarily affect the entire organism regarding homeostasis and regulatory functions. Triggerinduced systemic inflammatory reactions and various pathological biochemical changes of the organism, that in turn lead to further chronification, are characteristic. Among those multisystemic diseases related to inflammatory processes are for example diabetes, rheumatic diseases, neurodegenerative diseases, such as Parkinson‘s disease or multiple sclerosis, chronic infections and inflammatory bowel diseases, cardiovascular diseases, and allergies. All conditions mentioned above are on a rise in those countries predominantly following a western lifestyle.
Chronic inflammations do not only entail the activation of immune cells!
It is especially thanks to Martin L. Pall, Professor of biochemistry at Washington State University, that we are aware of the central role played by immunological and biochemical inflammatory phenomena in multisystemic diseases. Silent inflammations‘ characteristic elements are: continuous activation of inflammatory cells, nitrosative stress, oxidative stress, and acquired mitochondriopathy. Figure 1, based on Martin Pall‘s findings and modified to focus on immunological processes, depicts how those four elements play their part in a vicious circle of multisystemic diseases, since they mutually reinforce each other. Therefore it comes as no surprise when laboratory results often show that some or even all four regulatory compartments are affected.
The interrelation between mitochondriopathy and systemic inflammation is the most obvious. It explains the common finding in laboratory results regarding a decrease of intracellular ATP that coincides with increased inflammatory markers in blood (TNF-α or IP-10). Patients‘ individual genetic make-up and different patterns of interacting triggering factors explain exceptions from the rule, such as isolated disorders of individual compartments, especially at an early stage.
Every trigger sets off something different
Infectious agents, such as borrelia, chlamydia, herpes viruses and fungi, primarily activate inflammatory cells. In infections, nitrosative and oxidative stress as well as mitochondriopathy are rather secondary effects. Pollutants, such as metals, acrylates and solvents, have two ways of becoming triggering factors. For one, by means of immune cells‘ activation, especially if individual allergic sensitisations are already present, or dose-dependently in context with toxicological influences. Since the latter is often accompanied by malfunctioning cellular enzymatic and regulatory functions, toxicological effects commonly start with oxidative or nitrosative stress and disturbed mitochondrial function, while markers for immune system activation (still) demonstrate normal levels.
In summary, the pattern of four compartment markers‘ does at least in some cases allow for a differential diagnosis regarding individually responsible triggering factors. Since some or even all compartments may be affected during the chronic stage, since compartments may react individually and distinctly, and since in reality we are often met with multiple stressors, meaning multiple triggering factors, an unequivocal diagnosis is not always possible.
Chronic inflammations impart immune tolerance
Figure 1 shows how chronic inflammations impair the cellular immune system‘s ability to maintain immune tolerance. This explains how, as a result of a chronic systemic inflammation, triggers, which hitherto had been tolerated and until then never induced an activation of the immune system, become relevant as stimuli.
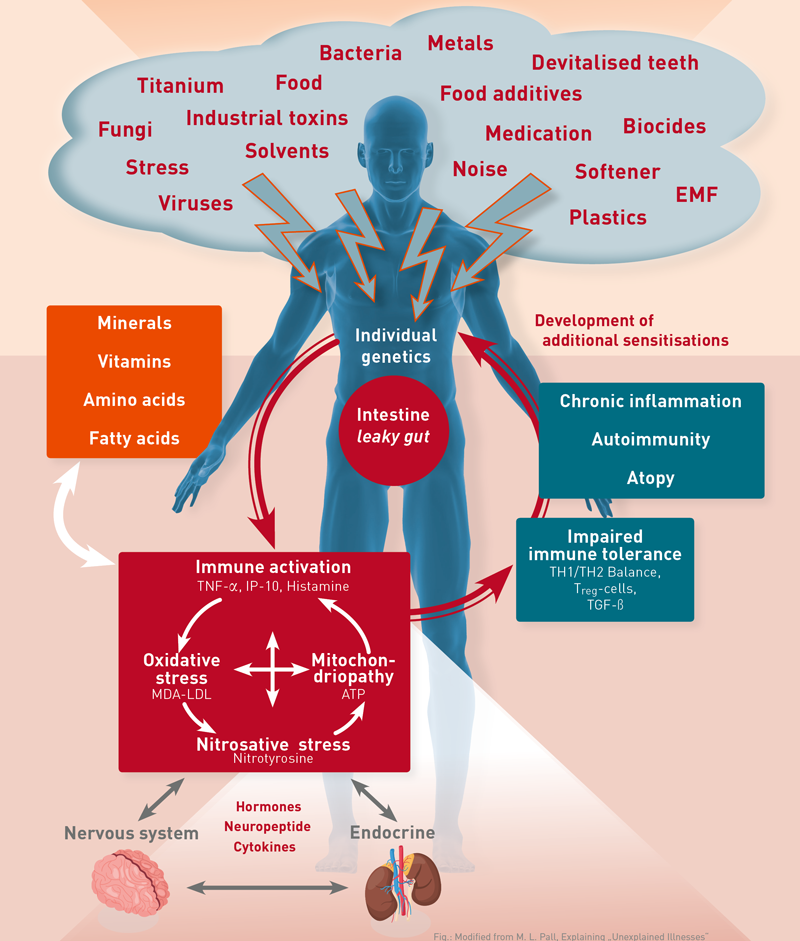
Fig. 1 see Martin L. Pall: Explaining “Unexplained Illnesses”: Disease Paradigm for Chronic Fatigue Syndrome, Multiple Chemical Sensitivity, Fibromyalgia, Posttraumatic Stress Disorder, Gulf War Syndrome and Others s‘:Disease Paradigm for Chronic Fatigue Syndrome, Multiple Chemical Sensitivity, Fibromyalgia, Posttraumatic Stress Disorder, Gulf War Syndrome and Others, ISBN 078902389X
Six laboratory markers are characteristic for inflammatory multisystemic diseases
TNF-α ➙ marker of myelomonocytic inflammations
TNF-α is a cytokine that is primarily secreted by macrophages. Due to its position at the start of the NFkB-associated inflammatory cascade TNF-α➙ IL1 ➙ IL6 ➙ IL10 ➙ IL1RA ➙ CRP, it has been labelled a pro-inflammatory key cytokine. Its prominent position makes it the most sensitive marker cytokine for myelomonocytic inflammations.
IP-10 ➙ TH1 immune activation
Interferon-gamma-induced protein 10 is secreted by monocytes in response to IFN-γ. Since IP-10 is almost exclusively induced by IFN-γ and occurs in higher levels in blood than the interferons themselves, this chemokine is an ideal biomarker for the quantification of IFN-γ‘s biological activity and TH1-celluar immune activation.
Histamine ➙ inflammation associated with mast cells
Histamine is a tissue hormone produced by mast cells. Contrary to common belief, mast cells and histamine are not exclusively involved in type 1 allergies, but instead play a role in almost every immune reaction against external antigens and allergens, and even stress. Histamine operates via four different types of receptors (HR1-HR4), affects various or-gans, such as the stomach, lungs, vessels, brain and colon, and stimulates numerous inflammatory phenomena by means of leukocytes‘ histamine receptors. The analysis is performed in heparinized whole blood, not in serum, since here stability is insufficient.
ATP (determined intracellularly) ➙ mitochondriopathy
ATP is produced by the respiratory chain in mitochondria. Therefore, ATP-levels determined in leukocytes are surrogate markers for current mitochondrial functions. Decreases very sensitively indicate an acquired mitochondriopathy.
MDA-LDL ➙ oxidative stress
Malondialdehyde-modified LDL (MDA-LDL) is an oxidised version of LDL-cholesterol, created by the reaction of lipids and oxygen radicals (lipid peroxidation). Hence, MDA-LDL is a biomarker of oxidative stress and is therefore, and in addition due to its longer half-life in serum, more suitable for an evaluation of oxidative stress than previously used pro-oxidative metabolic markers, such as malondialdehyde itself or enzyme activities, such as that of GlutathioneS-Transferase or Glutathione peroxidase.
Nitrotyrosine ➙ nitrosative stress = NO°/ONOO°-burden
Tyrosine is an amino acid occurring in blood. In the organism, nitrotyrosine is produced by reactive nitrogen species binding with tyrosine (nitrilation). Since this „transformation“ correlates with the burden of reactive nitrogen species, nitrotyrosine levels currently constitute the most stabile biomarker of nitrosative stress (more sensitive than citrulline in urine). A reference regarding total tyrosine levels in blood is not necessary, since nitrilation is associated with linear concentration ratios. Calculating quotients may, especially in connection with high tyrosine levels, lead to an underestimation of nitrosative stress.
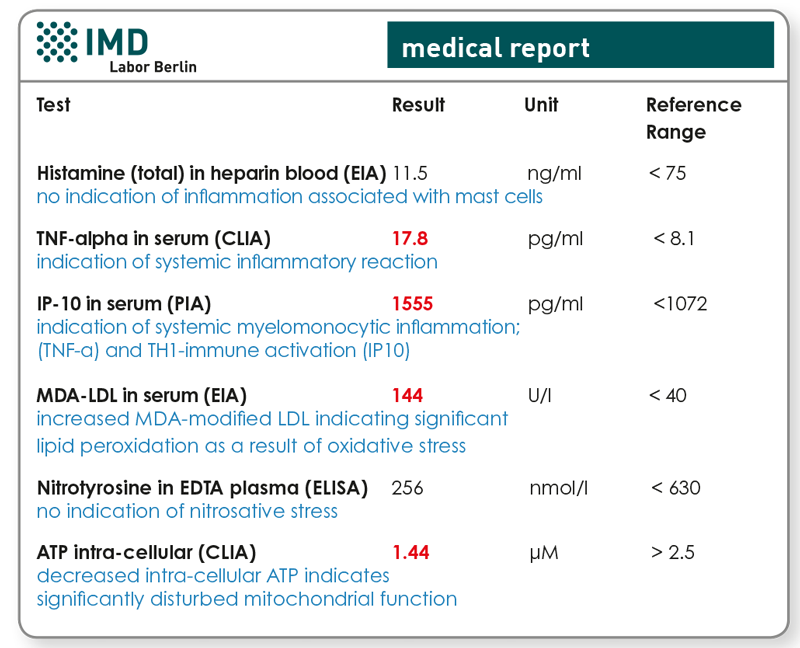
Indicators for the determination of the six presented laboratory markers
The sophisticated determination of those six laboratory markers allows for:
- status analysis of those four regulatory compartments indicating a chronic inflammatory multisystemic disease, for further ideal planning of substitutive therapy (antiinflammatory?, anti-oxidising?) and to monitor the therapy‘s progress
- differential diagnostics of individually responsible trig gering factors, at least in order to permit and narrow down cost-effective follow-up diagnostics (LTT, infection diagnostics, allergy diagnostics, toxicology, etc.) along side with the anamnesis.
Material
2x 10 ml heparin blood + 5 ml serum + 2.6 ml EDTA blood

