Lymphocyte transformation test (LTT)
What is the lymphocyte transformation test (LTT)?
LTT - history
Currently, the LTT is the only laboratory method to test for specific cellular sensitization.
First described in 1960, the test has evolved into a reproducible and highly sensitive method, taking advantage of the development in cell culture techniques and analytical methods.
Today its use extends beyond medical-biological research into routine diagnostic testing.
The test is based on the principle of antigen/allergen-specific induction of cell division in lymphocytes following contact with their “fitting” antigen.
A positive reaction in the LTT indicates the presence of antigen-specific lymphocytes (memory cells) in the patient’s blood.
What are the differences between today’s LTT and earlier versions of the test?
Around the year 2000, the LTT's sensitivity was still comparatively low. In type IV allergy testing, the LTT was at best equal to the skin test, if not inferior.
In these early years, its specificity in infection diagnostic testing was of concern.
It was relatively low as the quality of the test antigens available at that time was poor.
Frequently they triggered weakly positive and thus inconclusive reactions, making the interpretation of the results difficult.
This has changed dramatically in recent years.
Today, the LTT technologies used in specialised immunological laboratories are very reliable and offer high sensitivity and specificity.
LTT - importance / significance
Advances in cell culture methods and media, the quality of allergens and antigens used for cell stimulation and, last but not least, the fundamentally improved measuring methods available today have all contributed to this development.
The liquid scintillation counters used in the past to measure 3H-thymidine activity have been replaced by advanced solid-phase β counters, not requiring any subsequent manipulation of cultured cells.
Furthermore, the LTT now uses so-called micro-culture techniques which by allowing multiple test runs greatly enhance the reliability of the results.
Adding genetically engineered interferon-α to the cell culture has contributed to the development of this micro-culture technique among other modifications (von Baehr et al, J. Immunol. Methods 2001; 251: 63-71).
With the introduction of this optimised LTT version into routine testing in 2005, the test’s sensitivity and specificity greatly improved, especially in comparison with the MELISA methods that were still used at that time.
The lymphocyte transformation test is a demanding laboratory technology requiring not only expensive modern laboratory equipment but also laboratory staff with great experience and diligence. With the LTT, only few steps of the process can be automated. Even in today’s age of modern technology, good old manual processing by experienced staff trained in cell culture techniques is at the heart of cell cultures.
In addition, the level of standardisation in the respective laboratory has a significant impact on the analytical quality, as has the way how the methods were established and validated for each task. For this reason, cell-based analytic methods should only be used by specialized laboratories and institutes focussed on this technologies where these methods are accredited according to DIN 15189 by the National Accreditation Body for the Federal Republic of Germany (DAkkS).
The lymphocyte transformation test was first accredited in accordance with DIN 15189 at the Institut für Medizinische Diagnostik as early as 2004.
In its currently available modern version, the lymphocyte transformation test has firmly established itself as a medical diagnostic testing method for various diseases.
The LTT’s range of indications comprises 3 key areas of medical diagnostics:
- Immunodeficiency
Detection of deficiencies and functional impairments of the cellular immune system. See information. - Pathogens
Detection of the activity of chronic, persistent infections based on pathogen-specific T cell response (Borrelia, Chlamydia, Yersinia, Giardia lamblia, Herpes viruses, among others). See information. - Type IV allergy
Detection/exclusion of allergic sensitization of the cell-mediated type (type IV allergy) e.g. to medications, metals, acrylates, environmental pollutants, moulds or food allergens. See information.
LTT - procedure
Performing the LTT
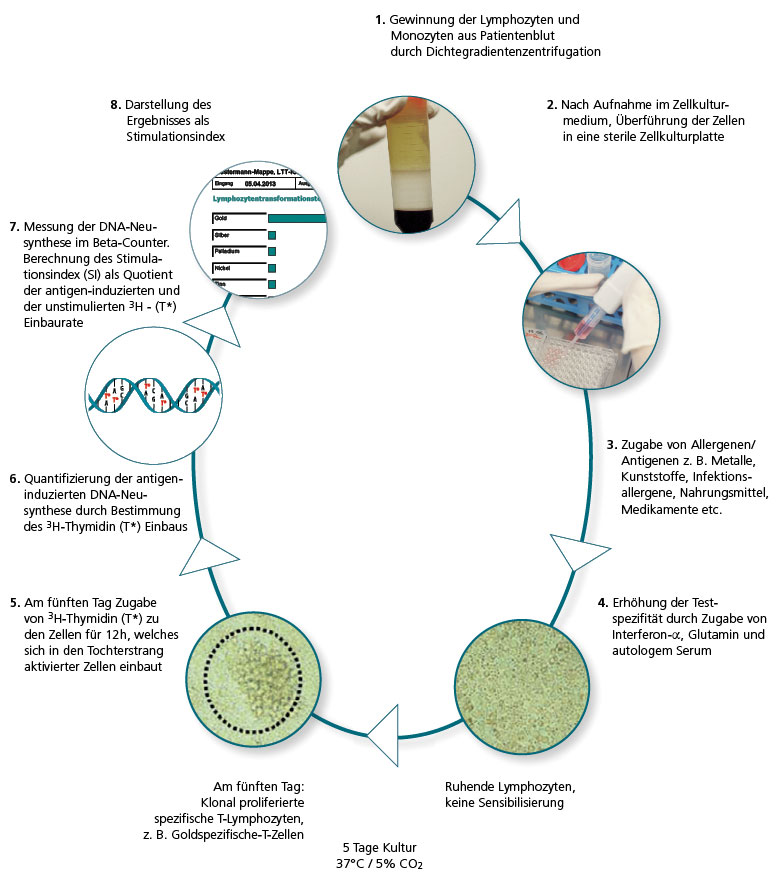
During the first step of the lymphocyte transformation test, the lymphocytes and monocytes (also referred to as mononuclear cells) are extracted by density gradient centrifugation using a Ficoll gradient.
Following several cleaning runs, the cells are collected in a special culture medium (with added IFN-α, among others) and then cultured.
The antigens to be tested are pipetted to the cells in triplicate and incubated under optimum growth conditions for 5 days. Subsequently, 3H-thymidine is added and the cells are cultured for further 12 hours.
During the phase of DNA synthesis, which only occurs in antigen/allergen-stimulated dividing lymphocytes, 3H-thymidine T* is integrated into the newly produced DNA strands and can then be quantified using a beta counter.
To show the result, the induced lymphocyte proliferation is calculated in comparison with the spontaneous proliferation (blank) and the result is reported as stimulation index (SI).
The stimulation index is the ratio of the proliferation rates in the stimulated test run and the non-stimulated blank.
LTT - analysis profile
The Institut für Medizinische Diagnostik offers the following LTT profiles:
Each of the antigens and allergens included in the profile can also be tested individually.
| Immune function testing | |
| LTT Immune Function | Testing of functional immune competence with antigens / mitogens Tetatoxoid, influenza, cytomegalovirus (CMV), varicella-zoster virus, streptokinase, candida, PWM |
| LTT Immune Stimulation | Test includes the LTT Immune Function (see above) PLUS the testing of immunomodulators at the requester’s choice (please enquire with the lab about the standardised preparations available) |
| Pathogen testing | |
| LTT-Borrelia | specific activity against recombinant OspC, B. sensu strictu, B. afzelii, B. garinii |
| LTT-Chlamydia trachomatis | specific activity against Chlamydia trachomatis antigen |
| LTT-Chlamydia pneumoniae | specific activity against Chlamydia pneumoniae antigen |
| LTT-Yersinia | specific activity against Yersinia enterocolitica antigen |
| LTT-Lamblia | specific activity against Giardia lamblia antigen |
| LTT-Helicobacter | specific activity against Helicobacter pylori antigen |
| LTT-Staphylococcus | specific activity against Staphylococcus aureus antigen |
| LTT-Streptococcus | specific activity against streptokinase |
| LTT-Candida | specific activity against Candida albicans antigen |
| LTT-Herpes viruses | CMV, EBV, VZV, HHV6, HSV1, HSV2 |
| Individual analyses | |
| LTT-HSV I | specific activity against Herpes simplex antigen |
| LTT-HSV II | specific activity against genital herpes antigen |
| LTT-VZV | specific activity against varicella-zoster antigen |
| LTT-CMV | specific activity against cytomegalovirus antigen |
| LTT-EBV | specific activity against Epstein-Barr virus antigen |
| LTT-HHV 6 | specific activity against human herpes virus-6 antigen |
| LTT-HHV 8 | specific activity against human herpes virus-8 antigen |
| Allergy Testing Drug allergy | |
| LTT Medications | detection of type IV sensitization (medications can be sent in as well) |
| LTT Ointment Bases | Petroleum jelly, lanolin, sorbiton, polyethylene glycol, triethanolamine, Eucerin, coconut oil, isopropyl myristate |
| Material intolerance | |
| LTT Dental Check | Metals: gold, nickel, palladium, chromium, cobalt, platinum, mercury, copper, silver, tin plastics: methyl methacrylate (MMA), hydroxyethyl methacrylate (HEMA), TEGDMA, BISGMA |
| LTT Metals | mercury, copper, silver, tin, ethylmercury, gold, nickel, palladium, chromium, cobalt, molybdenum, aluminium, platinum, cadmium |
| LTT Endoprosthetics | chromium, cobalt, molybdenum, nickel, titanium, vanadium, niobium, aluminium, zirconium (IV) oxide, methyl methacrylate, N,N-dimethyl-p-toluidine, benzoyl peroxide, hydroquinone, gentamicin |
| LTT Gold Alloy | gold, silver, platinum, copper, palladium, tin, gallium, indium, iridium, ruthenium, rhodium, tantalum |
| LTT Amalgam | mercury, copper, silver, tin, organic mercury compounds (ethyl-, phenyl- and methyl mercury) |
| LTT Plastics | TEGDMA, BISGMA, BISDMA, HEMA, methyl methacrylate (MMA), diurethane dimethacrylate (UDMA), ethylene glycol dimethacrylate, butanediol 1,4-methacrylate, hydroquinone, N,N-dimethyl-4-toluidine, benzoyl peroxide, formaldehyde, phthalate, camphorquinone |
| LTT Titanium | titanium, nickel, vanadium, aluminium |
| LTT Ceramics/Cements | vanadium, aluminium, titanium, cobalt, chromium, barium, silicon, cerium, boron, manganese, antimony, phosphate cement (Harvard), glass ionomer base cement (Ketac-Bond) |
| LTT Root Canal Filling Materials | raw gutta-percha, Balsam of Peru, eugenol, PDMS, silicone oil, bismuth oxide, silver, turpentine oil, colophony, triethanolamine, peanut oil, paraformaldehyde, bisphenol A, epichlorohydrin |
| LTT Native Materials | Testing of co-supplied materials which are prepared in the laboratory for analysis (e.g. material samples, cements, cosmetic products, solvent mixtures, etc.) |
| Environmental Pollutants / Moulds | |
| LTT MCS Environmental Factors | nickel, mercury, latex, PCP, PCB, permethrin, formaldehyde, methyl methacrylate, Aspergillus fumigatus, Penicillium chrysogenum, phthalic anhydride, dichlofluanid, PAK mix, 1,6-diisocyanatohexane |
| LTT Environmental Pollutants | formaldehyde, BTX, CKW, lindane, PAK mix, PCB, PCP, permethrin, latex, 1,6-diisocyanatohexane, phthalic anhydride, dichlofluanid |
| LTT Flame Retardants | Tris(2-chloroethyl) phosphate (TCEP), tris(2-butoxyethyl) phosphate (TBEP), tris(2-ethylhexyl) phosphate (TEHP) |
| LTT Plasticisers | Phthalic anhydride, diethyl phthalate, dimethyl phthalate, dibutyl phthalate, dioctyl phthalate |
| LTT Moulds | Aspergillus fumigatus, Penicillium chrysogenum, Trichophyton mentagrophytes, Cladosporum herbarum, Mucor mucedo, Rhizopus nigricans, Stachybotris spp., Botrytis cinerea, Alternaria alternate, Candida albicans (yeast) |
| LTT Beryllium | beryllium |
| Food Intolerance | |
| LTT Food screening 74 food item group testing + nickel | Meat: pig, chicken, beef, turkey, mutton (lamb), goose, duck Fish: trout, eel, halibut, salmon, cod, carp, tuna, herring Seafood: lobster, shrimp, crayfish, sole Cereals: wheat, barley, rye, oat, rice, spelt, corn, soy Brain food: Black tea, hops, cocoa beans, coffee beans, brewer's yeast Milk: milk protein, α-lactalbumine, β-lactoglobulin, casein, BSA (bovine serum albumin) Nuts: peanut, walnut, hazelnut, Brazil nut, pistachio, almond, cashew nut Garden fruits: apple, pear, strawberry, peach, grapes Citrus fruits: Orange, grapefruit, lemon, pineapple, mandarin, avocado, kiwi fruit, banana Spices: pepper, paprika, coriander, aniseed, cinnamon, vanilla, garlic Vegetables: pea, carrot, potato, cauliflower, celery, asparagus, spinach, tomato, onion Hen’s egg: egg yolk, egg white as well as baker's yeast as individually tested allergen and nickel as potent contact allergen |
| LTT Food TOP25 | cow’s milk, hen’s egg, wheat, rye, corn, spelt, carrot, potato, celery, spinach, tomato, apple, peach, orange, kiwi fruit, cod, tuna, beef, pork, chicken, paprika, hazelnut, peanut, baker’s yeast |
| LTT Gluten (gliadin) | Sensitisation to gliadin |