Autoantibodies against NEURONAL TRANSGLUTAMINASE 6 in suspected neurological manifestation of gluten intolerance
Gluten intolerance includes all conditions related to the consumption of foods containing gluten. These include coeliac disease (gluten-sensitive enteropathy), wheat allergy (allergic reactions to wheat components) and gluten sensitivity (all diagnosed disorders other than coeliac disease or allergy with intestinal and extraintestinal symptoms that occur after the consumption of foods containing gluten). Since gluten is ingested through food, gluten intolerance is expected to cause mostly gastroenterological symptoms. However, it has now become apparent that gluten intolerance can also manifest in other organs. These can include the skin (dermatitis herpetiformis Duhring) or the brain (e.g. gluten ataxia). In addition to the determination of auto-antibodies against tissue transglutaminase (anti-htTG/anti-TG2) or endomysium, biopsies can also be used to diagnose coeliac disease or dermatitis herpetiformis Duhring in the areas of gastroenterology and dermatology.
In the case of neurological diseases, however, biopsy is not an option.
Serological testing for antibodies against human neuronal transglutaminase 6 (TG6) opens up the possibility of identifying patients with an increased risk of neurological manifestations of gluten intolerance (Fig. 1).
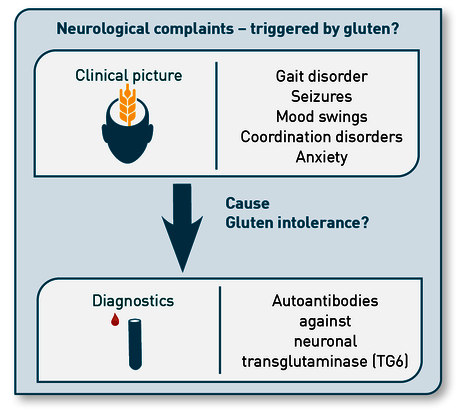
Fig. 1 Indications for the determination of TG6 autoantibodies
Pathogenesis
The pathogenesis of gluten-induced neurological conditions has not yet been fully clarified. Patients often exhibit abnormal cerebellar MR spectroscopy and white matter lesions or cerebellar atrophy.
Transglutaminase 6 (TG6), a member of the transglutaminase family of protein-crosslinking enzymes, is closely related to tissue transglutaminase 2 (TG2, the autoantigen in coeliac disease) and epidermal transglutaminase 3 (TG3, the autoantigen in dermatitis herpetiformis). TG6 is assumed to act as an autoantigen in gluten-related neurological diseases. Anti-TG6 IgA deposits have been observed in vessels in cerebellar tissue of patients with gluten ataxia. TG6 is expressed in the cerebellar cortex (in Purkinje cells), but also in the thalamus (motor control point). The significant thalamic atrophy and tendency to cerebellar atrophy observed in patients are therefore consistent with the loss of TG6 eurons, which may result in impairment of GABA-ergic inhibitory pathways.
TG6 in the diagnosis of gluten-associated diseases
TG6 autoantibodies in gluten-associated coeliac disease
Around 1/4 of adult patients with coeliac disease exhibit neurological symptoms. Anti-TG6 has been detected in up to 85% of coeliac patients with neurological involvement. On the one hand, this can be explained by a cross-reactivity of the TG2 autoantibodies with TG6; on the other hand, autoantibodies against other members of the transglutaminase family, in particular against TG6, also arise directly. In particular, patients not strictly adhering to a gluten-free diet develop increased amounts of antibodies against TG6. Failure to adhere to a gluten-free diet (GFD) increases the risk of a neurological manifestation of gluten intolerance. The risk increases with the duration of exposure to gluten. Analysis of TG6 autoantibodies is suitable for identifying coeliac patients at risk of neurological diseases and thus motivating them to adhere to a very strict GFD in order to prevent potential neurological comorbidities.
TG6 autoantibodies in gluten sensitivity (NCGS)
In addition to the guidelines, we (Diagnostic information 162) recommend primarily testing patients for autoantibodies against TG2 if coeliac disease is suspected. However, about a third of symptomatic patients who tested negative for TG2 formed isolated autoantibodies against other transglutaminases. The prevalence of positive TG6 autoantibodies is comparable between patients diagnosed with coeliac disease and NCGS. In this case, the documented neurological symptoms, which also occur at a comparable frequency, are also similar. The determination of autoantibodies against TG6 provides NCGS patients with a clear diagnosis and the possibility of targeted treatment through a strictly gluten-free
diet, and reduces the risk of neurological pathologies.
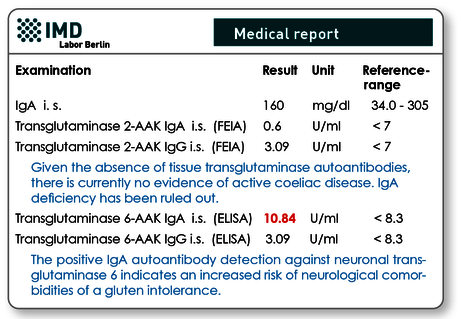
Fig. 2 Example of findings: Even in the absence of TG2-autoantibodies, the presence of TG6-autoantibodies indicates a gluten-induced autoimmune process.
TG6 autoantibodies for the diagnosis of gluten ataxia
Gluten ataxia (GA) accounts for 15% of all ataxias, particularly affects patients > 50 years of age and is characterised by a gradual onset of gait ataxia, associated with peripheral neuropathy. Similarly to paraneoplastic cerebellar degeneration, progression may be rapid in certain cases. The detection of auto-antibodies against TG6 is specific for GA in cases where the condition is suspected. Accordingly, the guideline for the diagnosis and treatment of progressive ataxia also recommends this examination as a sensitive marker for gluten-induced ataxia. In case of positive test results, a strictly gluten-free diet should be followed, even in the absence of enteropathy.
TG6 autoantibodies for the diagnosis of gluten neuropathy
In addition to gluten ataxia, gluten neuropathy may also occur. It manifests as a weakness of the extremities with numbness or tingling, up to the point of neuropathic pain in the hands and feet. Here, auto-antibodies against TG6 are used to diagnose gluten-induced neuropathy. In this case, too, a strictly gluten-free diet is indicated.
| Prevalence of TG6 autoantibodies in selected diseases | |
|---|---|
| Gluten ataxia | 73 % |
| Coeliac patients | 40 % |
| Coeliac patients with confirmed neurological involvement | 85 % |
| NCGS patients with neurological symptoms | 60 % |
| Cerebral palsy | 15 % |
| Schizophrenia (coeliac autoantibodies negative) | 13 % |
| Healthy individuals | < 4 % |
Differential diagnoses of treatable ataxias
In addition to gluten ataxia, other forms of treatable ataxias exist. Accordingly, diagnosis of these conditions is also important and should be taken into account in the context of differential diagnosis.
| Differential diagnoses of treatable ataxias | |
|---|---|
| Ataxia triggers | Diagnostics |
| Gluten | Transglutaminase 6 autoantibodies in serum |
| Vitamin B12 deficiency/ Autoimmune gastritis | Total vitamin B12, HoloTC, MMA, parietal cell/intrinsic factor autoantibodies in serum, homocysteine in citrate plasma (Diagnostic information 218) |
| Vitamin B1 deficiency/ alcoholism | B-vitamin status bioactive (Diagnostic information 307) |
| Vitamin E deficiency/ abetalipoproteinaemia | Vitamin E in serum (24H) LDL and VLDL in serum |
| Cerebrotendinous xanthomatosis | Serum cholestanol, CYP27A1 mutation |
| Niemann-Pick C disease (lipid storage disorder) | Examination of the cholesterol metabolism, Neurogenetic diagnostics investigation for NPC1 and NPC2 genes |
| Poisoning by alcohol, drugs (e.g. phenytoin), toxic metals (lead, mercury!) | Drug level, B-vitamin status bioactive (Diagnostic information 307), toxic metals in EDTA blood (Diagnostic information 304) |
| Infections | Syphilis, herpes zoster, Epstein-Barr virus, HIV, borreliosis |
| Paraneoplastic syndromes | Internal medicine diagnostics |
Material
neuronal transglutaminase 6 IgA and IgG autoantibodies: 1 ml of serum
Transport to the laboratory is not time-sensitive and can be sent by mail.
Invoicing
Please obtain the costs for the analysis from the pdf-document.
Literature
- Al-Toma A, Volta, Auricchio et al. European Society for the Study of Coeliac Disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. UEG J 2019 ; 7(5):583-613
- De Siva, Greenfield, Cook et al. Guidelines on the diagnosis and management of the progressive ataxias. Orphanet Journal of Rare Diseases 2019 ; 14:51
- Hadjivassiliou, Aeschlimann, Sanders et al. Transglutaminase 6 antibodies in the diagnosis of gluten ataxia. Neurology 2013 ; 80 (19):1740-5
- Hadjivassiliou, Rao, Grinewald et al. Neurological Dysfunction in Coeliac Disease and Non-Coeliac Gluten Sensitivity. Am J Gastroenterol 2016 ; 111(4):561-7
- Lionetti, Francavilla, Pavone et al. The neurology of coeliac disease in childhood: what is the evidence? A systematic review and meta-analysis. Dev Med child Neurol. 2010 ; 52:700-7007
- Zis, Rao, Sarrigiannis et al. Transglutaminase 6 antibodies in gluten neuropathy. Dig Liver Dis 2017 ; 49(11):1196-1200.
- Hadjivassiliou Croall Zis et al. Neurologic Deficits in Patients with Newly Diagnosed Celiac Disease Are Frequent and Linked With Autoantibodies to Transglutaminase 6. Clin Gastroenterol Hepatol. 2019 ; 17(13):2678- 2686
